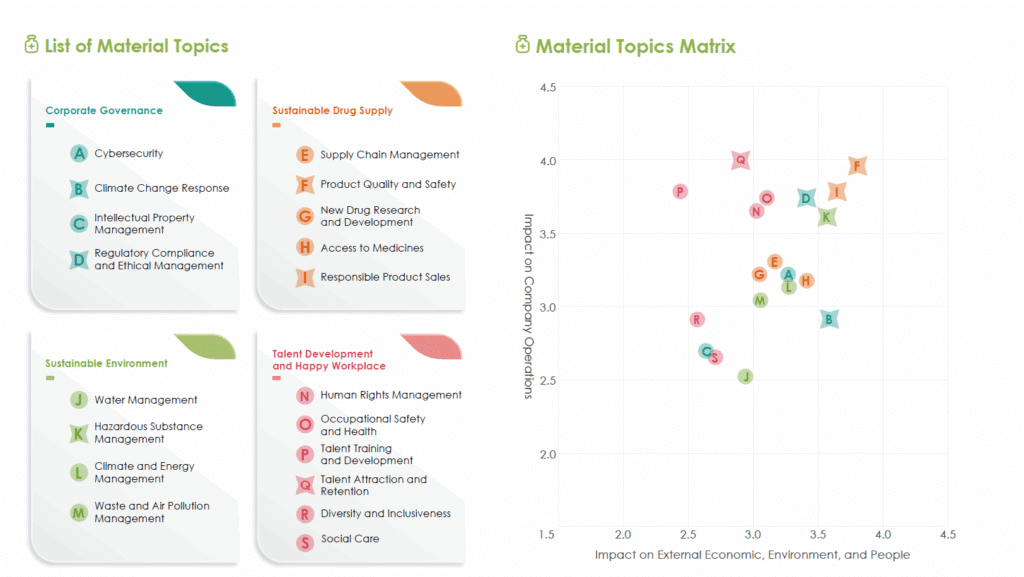
Material Topics Identification
Bora regularly collects sustainability concerns and trends through ongoing exchanges with stakeholders. Material Topics are updated based on feedback from previous sustainability reports and benchmarking against industry trends. Surveys are conducted with stakeholders and senior executives, and the feedback is analyzed and integrated.The results of this year’s material topics and the corresponding management approach were approved by the Board of Directors on May 14, 2025.
Material Topics and Management Guidelines
In 2023, Bora Pharmaceuticals identified 7 major topics through sustainable risk assessment processes. The relevant descriptions are outlined below.
| Aspect | Material Topic | Management Policies |
|---|---|---|
| Corporate Governance | Climate Change Response | Bora Pharmaceuticals fully recognizes the challenges and responsibilities brought by climate change. The Company is committed to taking actions to reduce greenhouse gas emissions and discloses governance, strategy, and risk management information in accordance with the TCFD framework. Every year, Bora conducts carbon inventories and formulates carbon reduction plans and measures to lower its operational carbon footprint. This approach meets market and customer expectations for carbon reduction targets while simultaneously strengthening corporate competitiveness. |
| Regulatory Compliance and Ethical Management | Bora Pharmaceuticals has implemented a comprehensive framework to ensure ethical conduct, including the “Sustainable Development Best Practice Principles,”“Code of Ethical Conduct,” and “Operating Procedures for Handling Material Inside Information and Prevention of Insider Trading”. These policies are overseen by the Corporate Governance Officer to promote accountability and compliance. In 2025, Bora began developing a group-wide risk control policy, which will be published on the Company’s website upon completion to enhance transparency and strengthen governance. | |
| Sustainable Drug Supply | Product Quality and Safety | All manufacturing sites strictly comply with international regulations including PIC/S GMP, US FDA CFR21, EU GMP, Health Canada, and Japan PMDA, and regularly undergo audits by regulatory authorities and customers. Additionally, the Company conducts internal self-inspections, with each quality system audited at least once annually to ensure continuous improvement and regulatory compliance. At the same time, Bora Pharmaceuticals has established and effectively implements a quality management system to ensure that all manufacturing and mass production operations meet GxP standards, relevant regulatory requirements, pharmacopeia standards, and product specifications, thereby maintaining high-quality product output. |
| Responsible Product Sales | The Company has established an internal review system and employee training mechanisms based on the pharmaceutical regulations of the product’s location and third-party organizational standards to ensure that all product promotion and sales activities comply with regulatory and ethical requirements. Additionally, through comprehensive complaint and whistleblowing channels, post-marketing safety monitoring, and regular audits of promotional materials, the Company continuously reviews and improves the compliance and transparency of its sales practices. | |
| Sustainable Environment | Hazardous Substance Management | The Company has implemented the “Chemical Cloud Management System” to integrate chemical information with geospatial information, effectively enhancing control over material flow from the source. For classification and storage, hazardous substances are managed with tiered controls and clear labeling, with specific storage regulations such as using explosion-proof cabinets for certain solvents and managing hazardous chemicals within fume hoods. Additionally, at least 2 chemical spill response drills are conducted annually, continuously strengthening the fire brigade’s awareness of hazardous material distribution within the facilities to ensure swift and proper emergency response. Furthermore, the Company proactively reports toxic chemical usage to regulatory authorities monthly and regularly undergoes audits by environmental agencies and the Science Park Administration to ensure compliance with relevant regulations. |
| Talent Development and Happy Workplace | Talent Attraction and Retention | The Company has established a fair and transparent talent attraction and retention mechanism through comprehensive recruitment and promotion systems, internal rotations, career planning, and key talent development programs, effectively enhancing employee satisfaction and sense of belonging. Additionally, the Company regularly participates in international salary surveys to ensure market-competitive compensation and offers a comprehensive and diverse benefits package, including performance bonuses, health insurance, and employee care programs. In terms of communication and employee engagement, the Company regularly holds labor management meetings and town hall meetings to actively listen to employee feedback and promote 2-way communication and interaction. Furthermore, an internal grievance mechanism is in place, complemented by the implementation of the third-party platform Conduct Watch as a whistleblowing channel to protect employee rights and maintain workplace fairness. |